|
8/13/2023 0 Comments Dendrite matrix We further show that PAR-6 is localized to the rosette vertex, and promotes DYF-7 localization, as well as the attachment to the epidermis and dendrite extension. 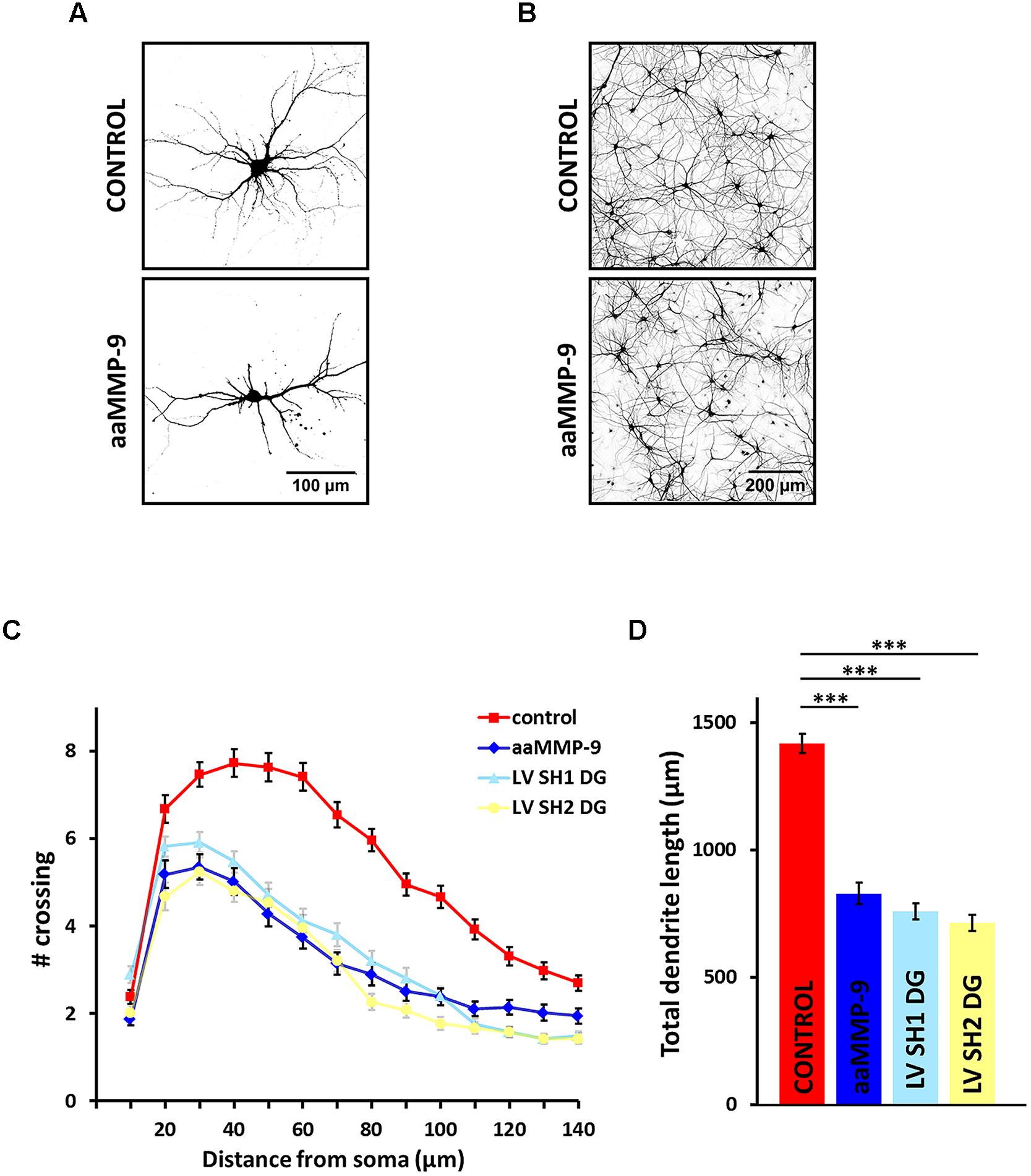
Molecular localization and loss of function phenotypes suggest that multiple adhesion molecules, including DYF-7, SAX-7/L1CAM, HMR-1/Cadherin and DLG-1/Dlg1 mediate attachment of the rosette vertex to the migrating epidermis. Abolishing epidermis migration by RNAi of elt-1, a key transcription factor required for epidermal fate, abolishes the extension of the amphid dendrites without affecting rosette formation. The vertex of the rosette, which becomes the dendrite tips, is attached to the anteriorly migrating epidermis and carried to the sensory depression at the developing nose, extruding the dendrites away from the neuronal cell bodies. Specifically, we show that the amphid neurons form a multicellular rosette along with the sheath and socket glial cells. Here, we report how amphid neurons orchestrate collective growth of their dendrites with morphogenesis of the surrounding epidermis. The anchoring requires DYF-7 and DEX-1, which likely assemble a matrix in the extracellular environment ( Heiman and Shaham, 2009). During development, these dendrites grow by retrograde extension, in which the dendritic tips are anchored in place at the embryonic nose while the cell bodies move posteriorly, extending the dendrite behind them ( Heiman and Shaham, 2009). This structure is highly organized, with the dendrites arranged in a stereotyped order within the bundle ( Yip and Heiman, 2018). The 12 dendrites in an amphid form a sensory nerve, which extends from the neck of the worm where the cell bodies are situated to the nose tip where most of the ciliated endings of the dendrites are exposed to the environment. Each amphid consists of 12 sensory neurons and two glia cells, namely the sheath and the socket cells. The amphids are a bilaterally symmetric pair of sensory organs in the nematode Caenorhabditis elegans. Multiple mechanisms coordinate the interaction between the pioneer and the followers, such as selective fasciculation ( Hayashi et al., 2014 Hutter, 2003 Lin et al., 1994), or juxtaparacrine signals ( Jaworski and Tessier-Lavigne, 2012). The follower neurons respond to cues from the pioneer ( Tamariz and Varela-Echavarría, 2015). In this model, the pioneer neuron extends its growth cone first to explore the environment and interact with chemotropic signals and environmental guideposts in order to establish a path to its target. 
The pioneer-follower model offers a simple developmental mechanism to explain this organization. In particular, many neurites bundle or fasciculate to form nerves. The guided outgrowth of neurites needs to be coordinated with other neurons as well as surrounding tissues ( Chao et al., 2009 Dong et al., 2015 Lefebvre et al., 2015). In the nervous system, sensory neurons may grow long dendrites and integrate their sensory endings into target tissues. Morphogenesis of an organism involves long-range coordination among organs. Our results suggest a collective mechanism of neurite extension that is distinct from the classical pioneer-follower model and highlight the role of mechanical cues from surrounding tissues in shaping neurites. PAR-6 is localized to the rosette vertex and dendrite tips, and promotes DYF-7 localization and dendrite extension. Multiple adhesion molecules including DYF-7, SAX-7, HMR-1 and DLG-1 function redundantly in rosette-to-epidermis attachment. The vertex of the rosette, which becomes the dendrite tips, is attached to the anteriorly migrating epidermis and carried to the sensory depression, extruding the dendrites away from the neuronal cell bodies. The amphid neurons first assemble into a multicellular rosette. elegans sensory organ, called the amphid, undergo a collective dendrite extension to form the sensory nerve. Coordination of neurite morphogenesis with surrounding tissues is crucial to the establishment of neural circuits, but the underlying cellular and molecular mechanisms remain poorly understood. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
